The clock is ticking. As of March 1, 2026, CMS will cease printing and mailing all paper fee coupons and physical CLIA certificates as part of its permanent discontinuation of this process.
This is not a gradual phase-in, nor is it a pilot program. This is an absolute cutoff that will forever change how each laboratory in America complies with CLIA (Clinical Laboratory Improvement Amendments).
CLIA certification affects over 317,000 laboratories across the U.S., so if your laboratory processes any type of human specimen for diagnostic testing, screening, or treatment purposes, this change applies to you. The mandatory transition to digital-only CLIA management creates immediate operational risks that most laboratories are not equipped to manage.
The Regulatory Catalyst: Why Digital-Only CLIA Starts Now
CMS has worked towards this point for years. In 2026, the final step to achieve the CMS objective of having the nation use an electronic form of communication to share health information as well as digital administrative processes will occur. A paper based certification process for CLIA laboratories has remained largely unaltered since the early 1990s.
Beginning March 1, 2026, state survey agencies will only send laboratory certifications renewals, fee notifications and other compliance updates through electronic means. Laboratories will no longer receive physical mailings of paper fee coupons that they have received for decades. Physical Certificates will no longer be issued, nor can they be mailed, in the same manner as before.
This change affects every laboratory category:
- Certificate of Waiver (CoW) Facilities
- Provider-Performed Microscopy (PPM) Laboratories
- Certificate of Compliance Laboratories
- Certificate of Accreditation Facilities
There are two exceptions to this policy; States like Washington and New York are both exempt from the CLIA regulations. All other laboratories in the United States will be required to electronically comply with the regulations.
The Technical Mechanics: How Digital CLIA Actually Works
CMS has been moving toward this point for many years. In 2026, the agency’s efforts to promote electronic health data exchange and digital administrative systems will reach a major milestone as the traditional paper based system for obtaining CLIA certification is phased out completely.
Starting March 1, 2026, all communications from state survey agencies regarding renewal of laboratory certifications (including notification of fees), and all compliance update communications, will occur electronically. Laboratory personnel will no longer receive paper fee coupons by U.S. Mail; physical certificates will not be mailed via U.S. postal service, nor can they be renewed through such means.
This change affects every laboratory category:
- Certificate of Waiver (CoW) facilities
- Provider-Performed Microscopy (PPM) laboratories
- Certificate of Compliance laboratories
- Certificate of Accreditation facilities
The requirement for electronic communication with laboratories will apply to each of the 50 States. Although CLIA exempt states such as Washington and New York have their own separate regulations; for virtually all U.S. laboratories, becoming compliant electronically will be mandatory-not optional.
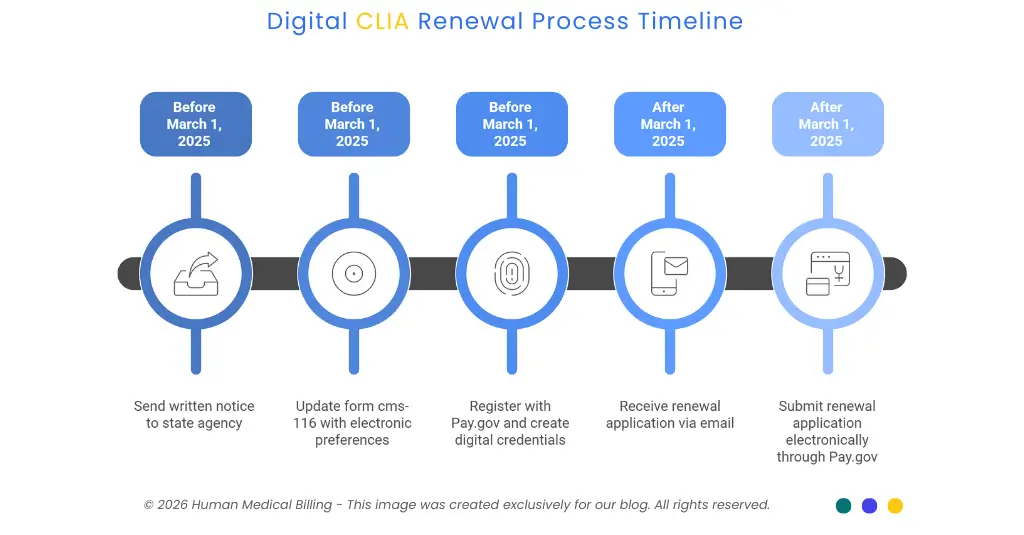
The transition requires laboratories to take three specific actions before the March deadline.
1. Step 1
Send a written notice to your state agency using an email to inform them of your intention to renew. You will have to provide your entire CLIA number, your Laboratory Director's Full Name, and an official signature. Your state agency is unable to accept telephone calls or verbal requests, nor can they accept a notification after March 1, 2025.
2. Step 2
Update form cms-116. This new version of the form will have a specific checkbox for electronic notification preferences and laboratories must complete and submit the updated form to activate digital communications. If you fail to check this box, you will remain in the old system until it shuts down completly.
3. Step 3
Register with Pay.gov as your laboratory will be required to make all future CLIA fees electronically through this federal payment system. As part of registering for Pay.gov, your laboratory will also have to create digital credentials to authenticate the identity of users logging into the system.
After the deadline, all laboratories will be sent a renewal application to their email account. The cost of renewal will remain the same; however, the way that your laboratory submits your renewal application will be changed to only being accepted electronically through Pay.gov.
In addition to having the capability to receive and send emails, there are several additional technical requirements that laboratories will be expected to meet in order to successfully submit their renewal application electronically.
Some of the additional technical requirements include:
- A dedicated business email address monitored daily
- Secure password management for Pay.gov and Login.gov accounts
- Calendar systems to track electronic renewal deadlines
- Digital signature capabilities for official documents
- Staff training on electronic submission protocols
The "Technical Denial" Risk: When Certification Lapses Hit Revenue
This is not a manual review process; it is an automated check by clearing houses and payers through comparison to the CLIA registry. When a CLIA certificate becomes inactive, automatic denial codes are triggered on claims submitted by laboratories, such as CO-109 (the claim is not covered by this payer/contractor), or CO-16 (the claim does not contain sufficient information or contains incorrect submission data).
CLIA numbers are reported on the CMS-1500 (Item 23) and also on Loop 2300 for electronic 837P submissions. Payors check the active status of a lab's certification prior to processing a claim. Anytime a CLIA certificate expires or becomes inactive, automatic front-end edits will deny claims immediately.
The financial cascade unfolds rapidly:
A missed $150 renewal fee causes labs to receive immediate denials for claims from all payers. A lab that performs about 200 tests each month, with an average reimbursement of $45/test would experience denied revenues of approximately $9000 within 30 days. Large reference labs perform tens of thousands of tests per day, resulting in denials that may reach hundreds of thousands of dollars in less than one week.
Accounts receivable days increase immediately. Clean claim ratios decrease dramatically. Lab staff devote time away from regular activities to manage the emergency of the situation. Credentialing departments are overwhelmed attempting to correct the payer enrollment files. The entire process of managing the revenue cycle in healthcare comes to a standstill due to what seems like an innocuous administrative error.
The recovery timeline adds to the problem as well. Once laboratories have obtained their CLIA certification again, resubmitting claims for payment can take a minimum of 30-45 days. Payers will accept corrected claims that include proof of an active laboratory certification in order to make payments. Some payers also request an attestation letter by Laboratory Directors. This creates a burden on the laboratories administrative function that exceeds the original missed deadline.
Operational Impact Study: Large Labs vs. Small Facilities
The digital mandate affects different laboratory types in strikingly different ways.
Large reference laboratories are generally well-equipped to meet the new digital requirements due to their established compliance departments and robust information technology (IT) infrastructures; as such, they are able to track renewals systematically and utilize existing electronic document management systems and federal payment portal experiences. Therefore, the digital mandate is essentially an improvement upon existing processes for these large reference laboratories and does not represent a major change in how they operate.
Physician Office Laboratories (POL) have an even tougher time than larger hospital based labs because they are typically run by a very few people and therefore do not have much in the way of administrative support. Often the Lab Director is a practicing physician who spends 90% of his or her time on actual patient care and therefore has little if any time for administrative duties such as keeping track of due dates for compliance related tasks and scheduling. Therefore when these offices are required to switch from a paper system of reminders to using a digital notification system, it will create additional workload for many small practices which may not be able to handle this new process.
The laboratory in a nursing home poses the greatest risk for compliance because most are classified as Certificate of Waivers under CLIA; thus, they represent a large majority of the approximately 80% of all CLIA certified labs that have waived their certification requirements. Additionally, these facilities frequently experience high employee turnover rates, little to no information technology (IT) support, and little or no administrative oversight. A single missed e-mail notice can cause a compliance failure at one facility that will then cause compliance failure at many other service lines in addition to the first.
In addition to the issues of the types of facilities in which laboratories are located, there are also location-based issues that impact compliance. Laboratories in rural locations often do not have adequate internet connection; therefore, they cannot connect reliably to federal payment portals. Thus, the disparate compliance risks created by the lack of technological resources available to underserved populations disproportionately negatively impacts those patients who are already in need of assistance.
The 5-Step Transition Roadmap: Your Implementation Checklist
Laboratories need to use a systematic approach in order to meet the March 1 deadline for digital transition of CLIA registration. Here is an example of how your lab can establish an operational protocol.
1. Step 1: verify your current CLIA information
Check the current CLIA certificate and confirm that all information listed on it is accurate. Check the name of the Laboratory director, facility address and contact details for each staff member. Any errors in the baseline data will cause cascading problems throughout the digital transition process. Immediately contact your state survey agency if there are any changes or corrections required to the information.
2. Step 2: Establish Digital Communication Channels
Create a specific business email address for CLIA related communications within your facility. Do not use personal email accounts or general info@ addresses used by multiple members of staff without coordination. Set up forwarding rules for the email address so at least two administrators receive all message from the state survey agency. Also configure calendar reminders for renewal deadlines which will be based on your facilities type of certification and renewal cycle.
3. Step 3: Set Up Your Account at Pay.gov
You will need to visit Pay.gov to set up your account through your facility’s Employer Identification Number (EIN) and link it to Login.gov for authentication purposes. Test the ability to pay electronically by making a low dollar transaction before you miss your renewal deadline. Store your login information in your facility's password management program that is secure.
4. Step 4: Submit State Agency Notification
Create your electronic notice email containing all of the required information (your facility's complete CLIA number; Laboratory Director's full name and credentials; facility name and address; Clearly stated request for all future communication via electronic means). Attach a digitally signed letter from your facility on official letterhead. Send this notification to your state survey agency's designated CLIA email address. Ask for verification of receipt and store the verification document in your compliance files.
5. Step 5: Update Form CMS-116
Complete the most recent version of Form CMS-116 that you downloaded from the CMS website; be sure to complete all required fields and check the electronic notification preference option. Once completed, submit it as outlined by your state agency (i.e., their process). Follow-up confirmation of receipt and processing within 10 business days.
Record all actions taken in your facility’s Compliance file(s); assign a specific individual or individuals at your facility responsibility for each of the action items identified; create backup processes for times when key personnel will be absent during peak renewal timeframes.
As laboratories transition through this, partnering with specialists in medical credentialing services may provide additional support in meeting compliance deadlines, so that operational focus is maintained on providing quality patient care.
The Future of Digital Oversight: Beyond CLIA Compliance
This is an example of a mandatory "digital shift" in CMS's broader strategy for both health care data exchange and administrative simplification. In its Interoperability and Patient Access Final Rule, CMS has required all health care entities to implement standardized electronic data-sharing methodologies. Therefore, the CLIA Paperless Mandate is entirely consistent with these two mandates.
In the near-term, anticipate CMS will be requiring other areas of regulation to move from a "paper-based" to a "digital-only" model. These would include; provider enrollment, Medicare certification and quality reporting programs. The CLIA transition will serve as a test-bed for how CMS will accomplish the transition of existing "legacy" paper-based systems to totally electronic operations.
State survey agencies are transitioning their regulatory compliance monitoring processes to electronic. Electronic Certificate Management allows State Survey Agencies (SSAs) to electronically verify the current status of laboratories in real-time. As such, payers can now electronically query a certification database instantaneously versus having to wait for paper documentation to be submitted. This provides a much more transparent regulatory environment and significantly increases the speed at which non-compliance becomes visible across the entire health care system.
The transition to a digital model presents laboratories with opportunities to modernize their own compliance operations. Laboratories may use digital document management, automated renewal tracking and/or electronic payment systems to reduce their long-term administrative burdens. Those facilities that begin to take advantage of the many compliance efficiencies that are provided by embracing this type of change will have established a position of readiness to meet future regulatory updates as opposed to constantly being reactive to each new deadline.
Preparing for March 1: The Action Timeline
Facilities must complete their digital transition by February 15, 2026 at the latest. This gives labs a two-week cushion from the deadline date. Waiting until the very end creates the opportunity for both technical issues and administrative mistakes which can cause compliance violations.
The transition is especially crucial for those with large-scale denial management services or large volumes of medical accounts receivable service. A small interruption in certification status can lead to delayed claims for months.
Review your facility's certificate renewal schedule today. All certificates issued in 2024 are due for renewal in 2026. Don't rely solely on how recently your last renewal occurred as the paperless requirement takes place regardless of when it is your next renewal cycle occurs.
If you need additional information regarding the transition process, contact your state survey agency. Many states have designated CLIA Coordinators which may be able to assist you with the e-submission process. Contact them first before relying solely on your interpretation of federal regulation.
You don't have to handle all the compliance burden yourself. There are partners such as Human Medical Billing that can help with compliance so you can focus on your core clinical operation. They also offer medical billing services and medical coding services to ensure that your claims continue to be processed correctly and efficiently through the compliance transition.
The Bottom Line: Digital or Denied
By March 1st 2026, the CLIA deadline will be final. The paper-based management of CLIA certifications will end completely by then. Laboratories that do not make the switch to digital management will have their certifications terminated. Any claims filed after the deadline will automatically be denied; resulting in a failure of all the revenue cycles.
There are only two possible outcomes here. Either the laboratory makes the transition to digital management before the deadline and will continue to operate without interruption, or the laboratory misses the deadline and will face immediate economic hardship as a result of denied claims.
It is clearly time to take action. If you don’t take action soon, you will have to pay later. Compliance costs include hours of administration work, while the cost of non-compliance includes lost revenue due to denied claims, disruptions in patient care, and potentially fines from the regulatory agencies.
Start your transition today. Update your CLIA information digitally; create a method to communicate with patients electronically; apply for a Pay.gov account; contact your State Agency (if applicable); and update your Form CMS-116. These are the required actions that will keep your laboratory compliant as well as ensure your revenue cycle continues to run smoothly in this new digital regulatory environment of healthcare.
The paper-based era of CLIA certification will come to an end in approximately three weeks. Ensure that your laboratory is prepared and does not fall behind.
If you need assistance with CLIA compliance, claims processing, and/or optimizing your laboratory’s revenue cycle, review Human Medical Billing’s specialized services designed specifically for laboratories throughout the United States. To learn more about Human Medical Billing’s services, review our services overview or contact us to discuss your laboratory’s unique needs.


